Power and Refrigeration Vapor Cycles: The Vapor Refrigeration Cycle and The Heat Pump
6.6 The Vapor Refrigeration Cycle
It is possible to extract heat from a space by operating a vapor cycle, similar to the Rankine cycle, in reverse. Work input is required in the operation of such a cycle, as shown in Fig. 6.10a. The work is input by a compressor that increases the pressure, and thereby the temperature, through an isentropic compression in the ideal cycle. The working fluid (often, R134a) then enters a condenser in which heat is extracted, resulting in saturated liquid. The pressure is then reduced in an expansion process so that the fluid can be evaporated with the addition of heat from the refrigerated space.
The most efficient cycle, a Carnot cycle, is shown in Fig. 6.10b. There are, how- ever, two major drawbacks when an attempt is made to put such a cycle into actual operation. First, it is not advisable to compress the mixture of liquid and vapor as
Figure 6.10 The vapor refrigeration cycle.
represented by state 1 in Fig. 6.10b since the liquid droplets would cause damage. Second, it would be quite expensive to construct a device to be used in the expansion process that would be nearly isentropic (no losses allowed). It is much simpler to reduce the pressure irreversibly by using an expansion valve in which enthalpy remains constant, as shown by the dotted line in Fig. 6.10c. Even though this expansion process is characterized by losses, it is considered to be part of the “ideal” vapor refrigeration cycle. Because the expansion process is a nonequilibrium process, the area under the T-s diagram does not represent the net work input.
The performance of the refrigeration cycle, when used as a refrigerator, is measured by
When the cycle is used as a heat pump, the performance is measured by
The coefficient of performance (COP) can attain values of perhaps 5 for properly designed heat pumps and 4 for refrigerators.
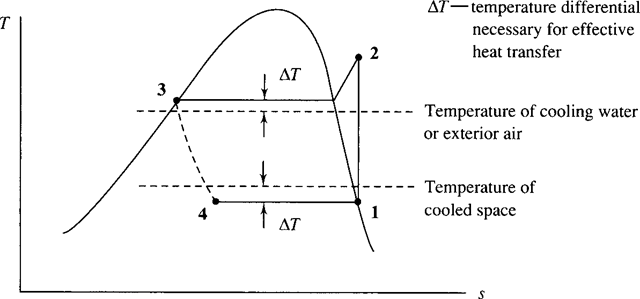
The condensation and evaporation temperatures, and hence the pressures, are established by the particular design of the refrigeration unit. For example, in a refrigerator that is designed to cool the freezer space to about −18°C (0°F), it is necessary to design the evaporator to operate at approximately −25°C to allow for effective
heat transfer between the space and the cooling coils. The refrigerant condenses by
transferring heat to air maintained at about 20°C; consequently, to allow for effective heat transfer from the coils that transport the refrigerant, the refrigerant must be maintained at a temperature of at least 27°C. This is shown in Fig. 6.11.
Figure 6.11 The refrigeration cycle showing design temperatures.
To accomplish refrigeration for most spaces, it is necessary that the evaporation temperature be quite low, in the neighborhood of −25°C, perhaps. This, of
course, rules out water as a possible refrigerant. Two common refrigerants are ammonia (NH ) and R134a. The thermodynamic properties of R134a are presented in App. D. The selection of a refrigerant depends on the high and low design temperatures. For example, temperatures well below −100°C are required to liquefy many gases. Neither ammonia nor R134a may be used at such low temperatures since they do not exist in a liquid form below −100°C. For most applications the refrigerant should be nontoxic, stable, environmentally benign, and relatively inexpensive.
Deviations from the ideal vapor refrigeration cycle include
• Pressure drops due to friction in connecting pipes.
• Heat transfer occurs from or to the refrigerant through the pipes connecting the components.
• Pressure drops occur through the condenser and evaporator tubes.
• Heat transfer occurs from the compressor.
• Frictional effects and flow separation occur on the compressor blades.
• The vapor entering the compressor may be slightly superheated.
• The temperature of the liquid exiting the condenser may be below the saturation temperature.
Quite often the size of an air conditioner is rated in tons, a rather interesting unit of rate of heat transfer. A “ton” of refrigeration is supposedly the heat rate necessary to melt a ton of ice in 24 hours. By definition, 1 ton of refrigeration equals 3.52 kW. Also, the performance of an air conditioner is often expressed as the EER (the Energy Efficiency Rating): the output in Btu’s divided by the energy purchased in kWh. We multiply the COP by 3.412 to obtain the EER.
EXAMPLE 6.9
R134a is used in an ideal vapor refrigeration cycle operating between saturation temperatures of −24°C in the evaporator and 39.39°C in the condenser. Calculate the rate of refrigeration, the coefficient of performance, and the rating in horsepower per ton if the refrigerant flows at 0.6 kg/s.
Solution
Refer to Fig. 6.10c. The enthalpy of each state is needed. From Table D.1 we find that h1 = 232.8 kJ/kg and s1 = 0.9370 kJ/kg ⋅ K, and from Table D.2 we find h3 = h4 = 105.3 kJ/kg. Using s1 = s2, we interpolate in Table D.3 at a pressure of 1.0 MPa (the pressure associated with the saturation temperature of 39.39 °C), and find that
The rate of refrigeration is measured by the heat transfer rate needed in the evaporation process, namely,
The power needed to operate the compressor is
The coefficient of performance is then calculated to be
The horsepower per ton of refrigeration is determined, with the appropriate con- version of units, as follows:
EXAMPLE 6.10
The ideal refrigeration cycle of Example 6.9 is used in the operation of an actual refrigerator. It experiences the following real effects:
• The refrigerant leaving the evaporator is superheated to −20°C.
• The refrigerant leaving the condenser is subcooled to 40°C.
• The compressor is 80 percent efficient.
Calculate the actual rate of refrigeration and the coefficient of performance.
Solution
From Table D.1 we find, using T = 40°C, that h3 = h4 = 106.2 kJ/kg. Also, from Table D.l we observe that P = 0.102 MPa. From Table D.3, at P1 = 0.10 MPa (close enough) and T = −20°C,
h1 = 236.5 kJ/kg s1 = 0.960 kJ/kg ⋅ K
If the compressor were isentropic, then, with s = s1 and P = 1.0 MPa,
From the definition of efficiency, η = ws /wa , we have
The rate of refrigeration is
Q·E = m· (h1 − h4 ) = 0.6 × (236.5 − 106.2) = 78.2 kJ/s
Note that the real effects have actually increased the capability to refrigerate a space. The coefficient of performance becomes
The 42 percent decrease in the COP occurs because the power input to the
compressor has increased substantially. But, observe that the refrigeration has increased.
6.7 The Heat Pump
The heat pump utilizes the vapor refrigeration cycle discussed in Sec. 6.6. It can be used to heat a house in cool weather or cool a house in warm weather, as shown schematically in Fig. 6.12. Note that in the heating mode the house gains heat from the condenser, whereas in the cooling mode the house loses heat to the evaporator.
Figure 6.12 The heat pump. (a) Heating. (b) Cooling.
This is possible since the evaporator and the condenser are similar heat exchangers and valves are used to perform the desired switching.
A heat pump system is sized to meet the heating load or the cooling load, which- ever is greater. In southern areas where the cooling loads are extremely large, the system may be oversized for the small heating demand of a chilly night; an air conditioner with an auxiliary heating system may be advisable in those cases. In a northern area where the large heating load demands a relatively large heat pump, the cooling load on a warm day may be too low for effective use of the heat pump; the large cooling capacity would quickly reduce the temperature of the house with- out a simultaneous reduction in the humidity, a necessary feature of any cooling system. In that case, a furnace that provides the heating with an auxiliary cooling system is usually advisable; or, the heat pump could be designed based on the cool- ing load, with an auxiliary heater for times of heavy heating demands.
EXAMPLE 6.11
A heat pump using R134a is proposed for heating a home that requires a maximum heating load of 300 kW. The evaporator operates at −12°C and the condenser at
800 kPa. Assume an ideal cycle.
(a) Determine the COP.
(b) Determine the cost of electricity at $0.07/kWh at peak load.
(c) Compare the R134a system with the cost of operating a furnace at peak load using natural gas at $0.50/therm if there are 100 000 kJ/therm of natural gas.
Solution
(a) Refer to the T-s diagram of Fig. 6.10c. From Table D.1 we find h1 = 240 kJ/kg, s1 = s2 = 0.927 kJ/kg · K, and h3 = h4 = 93.4 kJ/kg. Extrapolating at state 2 (P = 0.8 MPa), there results
The heat rejected by the condenser is
(b) Cost of electricity = 51.5 kW × $0.07/kWh = $3.60/h
(c) Assuming the gas furnace to be ideal, that is, it converts all of the energy Of the gas into usable heat, we have
The above gas furnace uses a heat pump that is assumed to operate ideally. A heat pump operates significantly below the ideal COP whereas a gas furnace can operate very near its ideal. The two systems are often quite competitive depend- ing on the cost of electricity compared with the price of gas.
Quiz No. 1
1. The component of the Rankine cycle that leads to a relatively low cycle efficiency is
(A) the pump
(B) the boiler
(C) the turbine
(D) the condenser
2. The pump in the Rankine cycle of Fig. 6.1 increases the pressure from
20 kPa to 6 MPa. The boiler exit temperature is 600°C and the quality
of the steam at the turbine exit is 100%. Find qboiler.
(A) 3410 kJ/kg
(B) 3070 kJ/kg
(C) 1050 kJ/kg
(D) 860 kJ/kg
3. Find wturbine for the Rankine cycle of Prob. 2.
(A) 3410 kJ/kg
(B) 3070 kJ/kg
(C) 1050 kJ/kg
(D) 860 kJ/kg
4. Find wpump for the Rankine cycle of Prob. 2.
(A) 4 kJ/kg
(B) 6 kJ/kg
(C) 8 kJ/kg
(D) 10 kJ/kg
5. The turbine efficiency of the turbine in Prob. 2 is nearest
(A) 64%
(B) 72%
(C) 76%
(D) 81%
6. The cooling water in the condenser of Prob. 2 is allowed a 10°C increase.
If m· steam = 2 kg/s, determine m· cooling water if saturated liquid leaves the
condenser.
(A) 80 kg/s
(B) 91 kg/s
(C) 102 kg/s
(D) 113 kg/s
7. The steam passing through the turbine of the power cycle of Prob. 2 is reheated at 800 kPa to 400°C. The thermal efficiency of the resulting cycle is nearest
(A) 33%
(B) 37%
(C) 40%
(D) 43%
8. If the turbine of Prob. 2 were 80 percent efficient, estimate the temperature that would exist at the turbine outlet.
(A) 60°C
(B) 65°C
(C) 70°C
(D) 75°C
9. An open feedwater heater is inserted into the cycle of Prob. 2 at 800 kPa.
Calculate the mass m5 in Fig. 6.5 if m6 = 1 kg.
(A) 0.170 kg
(B) 0.192 kg
(C) 0.221 kg
(D) 0.274 kg
10. An open feed water heater accepts superheated steam at 400 kPa and 200°C
and mixes the steam with condensate at 20 kPa from an ideal condenser.
If 20 kg/s flows out of the condenser, what is the mass flow rate of the superheated steam?
(A) 2.26 kg/s
(B) 2.71 kg/s
(C) 3.25 kg/s
(D) 3.86 kg/s
11. R134a is used in an ideal refrigeration cycle between pressures of 120 kPa and 1000 kPa. If the compressor requires 10 hp, calculate the rate of refrigeration.
(A) 15.9 kJ/s
(B) 17.2 kJ/s
(C) 21.8 kJ/s
(D) 24.4 kJ/s
12. The coefficient of performance for the refrigerator of Prob. 11 is nearest (A) 2.31
(B) 2.76
(C) 2.93
(D) 3.23
13. A heat pump using R134a as the refrigerant provides 80 MJ/h to a building.
The cycle operates between pressures of 1000 and 200 kPa. Assuming an ideal cycle the compressor horsepower is nearest
(A) 3.28
(B) 3.87
(C) 4.21
(D) 4.97
14. The volume flow rate into the compressor of the ideal heat pump of Prob. 13 is nearest
(A) 0.031 m3/s
(B) 0.026 m3/s
(C) 0.019 m3/s
(D) 0.013 m3/s
Quiz No. 2
1. The Rankine power cycle is idealized. Which of the following is not one of the idealizations?
(A) Friction is absent.
(B) Heat transfer does not occur across a finite temperature difference.
(C) Pressure drops in pipes are neglected.
(D) Pipes connecting components are insulated.
2. A steam power plant is designed to operate on a Rankine cycle with a condenser pressure of 100 kPa and boiler outlet temperature of 500°C. If the pump outlet pressure is 2 MPa, calculate the ideal turbine work output.
(A) 764 kJ/kg
(B) 723 kJ/kg
(C) 669 kJ/kg
(D) 621 kJ/kg
3. The boiler requirement of the cycle of Prob. 2 is nearest
(A) 2280 kJ/kg
(B) 2750 kJ/kg
(C) 2990 kJ/kg
(D) 3050 kJ/kg
4. The maximum possible thermal efficiency of the cycle of Prob. 2 is nearest
(A) 31%
(B) 28%
(C) 25%
(D) 23%
5. If m· = 2 kg/s for the cycle of Prob. 2, the pump horsepower would be
nearest
(A) 4.0 hp
(B) 5.1 hp
(C) 6.2 hp
(D) 7.0 hp
6. The steam passing through the turbine of the power cycle of Prob. 2 is reheated at 800 kPa to 500°C. The thermal efficiency of the resulting reheat Rankine cycle is nearest
(A) 26.2%
(B) 28.0%
(C) 28.8%
(D) 29.2%
7.An open feedwater heater is inserted into the Rankine cycle of Prob. 2 at
800 kPa. Calculate the mass min Fig. 6.5 if m = 1 kg.
(A) 0.14 kg
(B) 0.13 kg
(C) 0.12 kg
(D) 0.11 kg
8. The Rankine cycle of Prob. 2 with the feedwater heater of Prob. 7 (refer to Fig. 6.5) has a maximum efficiency nearest
(A) 24%
(B) 26%
(C) 28%
(D) 30%
9. If the turbine of Prob. 2 is 90 percent efficient, estimate the temperature at the turbine outlet.
(A) 152°C
(B) 154°C
(C) 156°C
(D) 158°C
10. Refrigerant flows through the coil on the back of most refrigerators. This
coil is the
(A) evaporator
(B) intercooler
(C) reheater
(D) condenser
11. An ideal vapor refrigeration cycle utilizes R134a as the working fluid
between saturation temperatures of −28 and 40°C. For a flow of 0.6 kg/s,
determine the rate of refrigeration.
(A) 68.3 kJ/s
(B) 70.2 kJ/s
(C) 74.5 kJ/s
(D) 76.9 kJ/s
12. The coefficient of performance for the refrigerator of Prob. 11 is nearest (A) 3.52
(B) 3.15
(C) 2.74
(D) 2.52
13. The compressor of a refrigeration cycle accepts R134a as saturated vapor at 200 kPa and compresses it to 1200 kPa. The R134a leaves the condenser at 40°C. Determine the mass flow rate of R134a for 10 tons of refrigeration.
(A) 0.261 kg/s
(B) 0.349 kg/s
(C) 0.432 kg/s
(D) 0.566 kg/s
14. Calculate the minimum compressor horsepower for the ideal heat pump of Prob. 13.
(A) 10.1 hp
(B) 11.8 hp
(C) 13.3 hp
(D) 15.2 hp
15. A heat pump uses groundwater at 12°C as an energy source. If the energy delivered by the heat pump is to be 60 MJ/h, estimate the minimum mass flow rate of groundwater if the compressor operates with R134a between pressures of 100 kPa and 1.0 MPa.
(A) 0.421 kg/s
(B) 0.332 kg/s
(C) 0.303 kg/s
(D) 0.287 kg/s