Chemical Effects of Electricity
Introduction
A material must contain charged particles to be able to conduct electric current. In solids, electrons carry the current. Copper, lead, aluminium, iron and carbon are some examples of solid conductors. In liquids and gases, the current is carried by the part of a molecule that has acquired an electric charge, called ions. These can possess a positive or negative charge, and examples include hydrogen ion HC, copper ion Cu++ and hydroxyl ion OH-. Distilled water contains no ions and is a poor conductor of electricity, whereas salt water contains ions and is a fairly good conductor of electricity.
Electrolysis
Electrolysis is the decomposition of a liquid compound by the passage of electric current through it. Practical applications of electrolysis include the electroplating of metals, the refining of copper and the extraction of aluminium from its ore.
An electrolyte is a compound that will undergo electrolysis. Examples include salt water, copper sulphate and sulphuric acid.
The electrodes are the two conductors carrying current to the electrolyte. The positive-connected electrode is called the anode and the negative-connected electrode the cathode.
When two copper wires connected to a battery are placed in a beaker containing a salt-water solution, current will flow through the solution. Air bubbles appear around the wires as the water is changed into hydrogen and oxygen by electrolysis.
Electroplating
Electroplating uses the principle of electrolysis to apply a thin coat of one metal to another metal. Some practical applications include the tin-plating of steel, silver-plating of nickel alloys and chromium plating of steel. If two copper electrodes connected to a battery are placed in a beaker containing copper sulphate as the electrolyte it is found that the cathode (i.e. the electrode connected to the negative terminal of the battery) gains copper whilst the anode loses copper.
The Simple Cell
The purpose of an electric cell is to convert chemical energy into electrical energy.
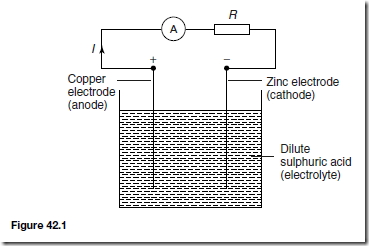
A simple cell comprises two dissimilar conductors (electrodes) in an electrolyte. Such a cell is shown in Figure 42.1, comprising copper and zinc electrodes. An electric current is found to flow between the electrodes. Other possible electrode pairs exist, including zinc-lead and zinc-iron. The electrode potential (i.e. the p.d. measured between the electrodes) varies for each pair of metals. By knowing the e.m.f. of each metal with respect to some standard electrode, the e.m.f. of any pair of metals may be determined. The standard used is the hydrogen electrode. The electrochemical series is a way of listing elements in order of electrical potential, and Table 42.1 shows a number of elements in such a series.
In a simple cell two faults exist — those due to polarisation and local action.
Polarisation
If the simple cell shown in Figure 42.1 is left connected for some time, the current I decreases fairly rapidly. This is because of the formation of a film of hydrogen bubbles on the copper anode. This effect is known as the polarisation of the cell. The hydrogen prevents full contact between the copper electrode and the electrolyte and this increases the internal resistance of the
cell. The effect can be overcome by using a chemical depolarising agent or depolariser, such as potassium dichromate that removes the hydrogen bubbles as they form. This allows the cell to deliver a steady current. For more on polarisation, see chapter 78.
Local action
When commercial zinc is placed in dilute sulphuric acid, hydrogen gas is liberated from it and the zinc dissolves. The reason for this is that impurities, such as traces of iron, are present in the zinc that set up small primary cells with the zinc. These small cells are short-circuited by the electrolyte, with the result that localised currents flow causing corrosion. This action is known as local action of the cell. This may be prevented by rubbing a small amount of mercury on the zinc surface, which forms a protective layer on the surface of the electrode.
When two metals are used in a simple cell the electrochemical series may be used to predict the behaviour of the cell:
(i) The metal that is higher in the series acts as the negative electrode, and vice-versa. For example, the zinc electrode in the cell shown in Figure 42.1 is negative and the copper electrode is positive.
(ii) The greater the separation in the series between the two metals the greater is the e.m.f. produced by the cell.
The electrochemical series is representative of the order of reactivity of the metals and their compounds:
(i) The higher metals in the series react more readily with oxygen and vice- versa.
(ii) When two metal electrodes are used in a simple cell the one that is higher in the series tends to dissolve in the electrolyte.
Corrosion
Corrosion is the gradual destruction of a metal in a damp atmosphere by means of simple cell action. In addition to the presence of moisture and air required for rusting, an electrolyte, an anode and a cathode are required for corrosion. Thus, if metals widely spaced in the electrochemical series, are used in contact with each other in the presence of an electrolyte, corrosion will occur. For example, if a brass valve is fitted to a heating system made of steel, corrosion will occur.
The effects of corrosion include the weakening of structures, the reduction of the life of components and materials, the wastage of materials and the expense of replacement.
Corrosion may be prevented by coating with paint, grease, plastic coatings and enamels, or by plating with tin or chromium. Also, iron may be galvanised, i.e. plated with zinc, the layer of zinc helping to prevent the iron from corroding.
E.m.f. and Internal Resistance of a Cell
The electromotive force (e.m.f.), E, of a cell is the p.d. between its terminals when it is not connected to a load (i.e. the cell is on ‘no load’).
The e.m.f. of a cell is measured by using a high resistance voltmeter connected in parallel with the cell. The voltmeter must have a high resistance otherwise it will pass current and the cell will not be on ‘no-load’. For example, if the resistance of a cell is 1 Q and that of a voltmeter 1 MQ then the equivalent resistance of the circuit is 1 MQ C 1 Q, i.e. approximately 1 MQ, hence no current flows and the cell is not loaded.
The voltage available at the terminals of a cell falls when a load is connected. This is caused by the internal resistance of the cell, that is, the opposition of the material of the cell to the flow of current. The internal resistance acts in series with other resistances in the circuit. Figure 42.2 shows a cell of e.m.f. E volts and internal resistance, r, and XY represents the terminals of the cell.
When a load (shown as resistance R) is not connected, no current flows and the terminal p.d., V D E. When R is connected a current I flows which causes a voltage drop in the cell, given by Ir. The p.d. available at the cell terminals is less than the e.m.f. of the cell and is given by:
Thus, if a battery of e.m.f. 12 volts and internal resistance 0.01 Q delivers a current of 100 A, the terminal p.d.,
V = 12 - (100)(0.01) = 12 - 1 = 11 V
When different values of potential difference V across a cell or power supply are measured for different values of current I, a graph may be plotted as shown in Figure 42.3. Since the e.m.f. E of the cell or power supply is the p.d. across its terminals on no load (i.e. when I D 0), then E is as shown by the broken line.
Since V = E - Ir then the internal resistance may be calculated from

When a current flows in the opposite direction to that shown in Figure 42.2 the cell is said to be charging (V > E).
A battery is a combination of more than one cell. The cells in a battery may be connected in series or in parallel.
(i) For cells connected in series:
Total e.m.f. = sum of cell’s e.m.f.’s
Total internal resistance = sum of cell’s internal resistance’s
(ii) For cells connected in parallel:
If each cell has the same e.m.f. and internal resistance: Total e.m.f. D e.m.f. of one cell
Total internal resistance of n cells
For example, eight cells, each with an internal resistance of 0.2 Q and an e.m.f. of 2.2 V are connected (a) in series, (b) in parallel.
Primary Cells
Primary cells cannot be recharged, that is, the conversion of chemical energy to electrical energy is irreversible and the cell cannot be used once the chemicals are exhausted. Examples of primary cells include the Leclanche´ cell and the mercury cell.
Lechlanche´ cell
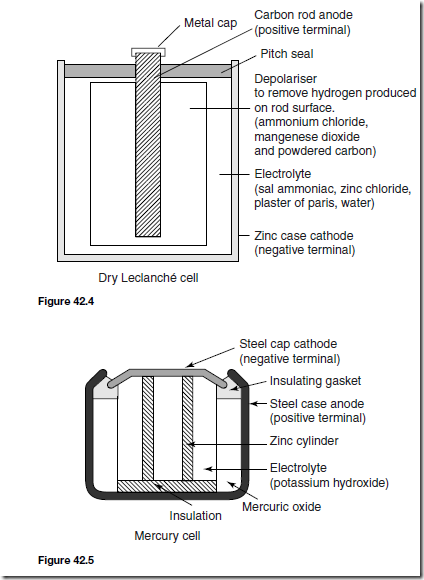
A typical dry Lechlanche´ cell is shown in Figure 42.4. Such a cell has an e.m.f. of about 1.5 V when new, but this falls rapidly if in continuous use due to polarisation. The hydrogen film on the carbon electrode forms faster than can be dissipated by the depolarizer. The Lechlanche´ cell is suitable only for intermittent use, applications including torches, transistor radios, bells, indicator circuits, gas lighters, controlling switch-gear, and so on. The cell is the most commonly used of primary cells, is cheap, requires little maintenance and has a shelf life of about 2 years.
Mercury cell
A typical mercury cell is shown in Figure 42.5. Such a cell has an e.m.f. of about 1.3 V which remains constant for a relatively long time. Their main advantage over the Lechlanche´ cell is its smaller size and its long shelf life. Typical practical applications include hearing aids, medical electronics, cam- eras and for guided missiles.
Secondary Cells
Secondary cells can be recharged after use, that is, the conversion of chemical energy to electrical energy is reversible and the cell may be used many times. Examples of secondary cells include the lead-acid cell and the alkaline cell. Practical applications of such cells include car batteries, telephone circuits and for traction purposes — such as milk delivery vans and fork lift trucks.
Lead-acid cell
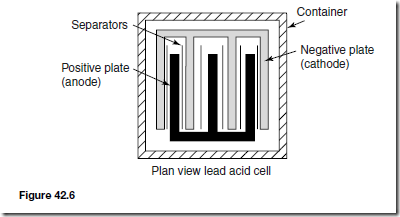
A typical lead-acid cell is constructed of:
(i) A container made of glass, ebonite or plastic.
(ii) Lead plates
(a) the negative plate (cathode) consists of spongy lead
(b) the positive plate (anode) is formed by pressing lead peroxide into the lead grid.
The plates are interleaved as shown in the plan view of Figure 42.6 to increase their effective cross-sectional area and to minimise internal resistance.
(iii) Separators made of glass, celluloid or wood.
(iv) An electrolyte which is a mixture of sulphuric acid and distilled water.
The relative density (or specific gravity) of a lead-acid cell, which may be measured using a hydrometer, varies between about 1.26 when the cell is fully charged to about 1.19 when discharged. The terminal p.d. of a lead-acid cell is about 2 V.
When a cell supplies current to a load it is said to be discharging. During discharge:
(i) the lead peroxide (positive plate) and the spongy lead (negative plate) are converted into lead sulphate, and
(ii) the oxygen in the lead peroxide combines with hydrogen in the electrolyte to form water. The electrolyte is therefore weakened and the relative density falls.
The terminal p.d. of a lead-acid cell when fully discharged is about 1.8 V. A cell is charged by connecting a d.c. supply to its terminals, the positive
terminal of the cell being connected to the positive terminal of the supply. The charging current flows in the reverse direction to the discharge current, and the chemical action is reversed. During charging:
(i) the lead sulphate on the positive and negative plates is converted back to lead peroxide and lead respectively, and (ii) the water content of the electrolyte decreases as the oxygen released from the electrolyte combines with the lead of the positive plate. The relative density of the electrolyte thus increases.
The colour of the positive plate when fully charged is dark brown and when discharged is light brown. The colour of the negative plate when fully charged is grey and when discharged is light grey.
Alkaline cell
There are two main types of alkaline cell — the nickel-iron cell and the nickel- cadmium cell. In both types the positive plate is made of nickel hydroxide enclosed in finely perforated steel tubes, the resistance being reduced by the addition of pure nickel or graphite. The tubes are assembled into nickel-steel plates.
In the nickel-iron cell, (sometimes called the Edison cell or nife cell), the negative plate is made of iron oxide, with the resistance being reduced by a little mercuric oxide, the whole being enclosed in perforated steel tubes and assembled in steel plates. In the nickel-cadmium cell the negative plate is made of cadmium. The electrolyte in each type of cell is a solution of potassium hydroxide that does not undergo any chemical change and thus the quantity can be reduced to a minimum. The plates are separated by insulating rods and assembled in steel containers that are then enclosed in a non-metallic crate to insulate the cells from one another. The average discharge p.d. of an alkaline cell is about 1.2 V.
Advantages of an alkaline cell (for example, a nickel-cadmium cell or a nickel-iron cell) over a lead-acid cell include:
(i) More robust construction
(ii) Capable of withstanding heavy charging and discharging currents without damage
(iii) Has a longer life
(iv) For a given capacity is lighter in weight
(v) Can be left indefinitely in any state of charge or discharge without damage
(vi) Is not self-discharging
Disadvantages of an alkaline cell over a lead-acid cell include:
(i) Is relatively more expensive
(ii) Requires more cells for a given e.m.f.
(iii) Has a higher internal resistance
(iv) Must be kept sealed
(v) Has a lower efficiency
Alkaline cells may be used in extremes of temperature, in conditions where vibration is experienced or where duties require long idle periods or heavy
discharge currents. Practical examples include traction and marine work, lighting in railway carriages, military portable radios and for starting diesel and petrol engines.
However, the lead-acid cell is the most common one in practical use.
Cell Capacity
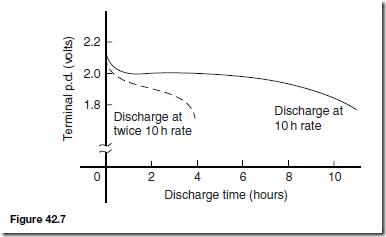
The capacity of a cell is measured in ampere-hours (Ah). A fully charged 50 battery rated for 10 h discharge can be discharged at a steady current of 5 A for 10 h, but if the load current is increased to 10 A then the battery is discharged in 3– 4 h, since the higher the discharge current, the lower is the effective capacity of the battery. Typical discharge characteristics for a lead-acid cell are shown in Figure 42.7.