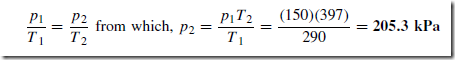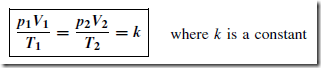Ideal gas Laws
Ideal gas Laws
Introduction
The relationships that exist between pressure, volume and temperature in a gas are given in a set of laws called the gas laws.
Boyle’s Law
Boyle’s law states:
the volume V of a fixed mass of gas is inversely proportional to its absolute pressure p at constant temperature
Charles’ Law
Charles’ law states:
for a given mass of gas at constant pressure, the volume V is directly proportional to its thermodynamic temperature T
A process that takes place at constant pressure is called an isobaric process.
The relationship between the Celsius scale of temperature and the thermodynamic or absolute scale is given by:
The Pressure Law
The pressure law states:
the pressure p of a fixed mass of gas is directly proportional to its thermodynamic temperature T at constant volume
When a fixed mass of gas at constant volume changes from pressure p1 and temperature T1, to pressure p2 and temperature T2 then:
For example, gas initially at a temperature of 17° C and pressure 150 kPa is heated at constant volume until its temperature is 124° C. Assuming no loss of gas, the final pressure of the gas is determined from:
Dalton’s Law of Partial Pressure
Dalton’s law of partial pressure states:
the total pressure of a mixture of gases occupying a given volume is equal to the sum of the pressures of each gas, considered separately, at constant temperature The pressure of each constituent gas when occupying a fixed volume alone is known as the partial pressure of that gas.
An ideal gas is one that completely obeys the gas laws given above. In practice no gas is an ideal gas, although air is very close to being one. For calculation purposes the difference between an ideal and an actual gas is very small.
For example, a gas R in a container exerts a pressure of 200 kPa at a temperature of 18°C. Gas Q is added to the container and the pressure increases to 320 kPa at the same temperature. The pressure that gas Q alone exerts at the same temperature, is given by Dalton’s law of partial pressure:
Characteristic Gas Equation
Frequently, when a gas is undergoing some change, the pressure, temperature and volume all vary simultaneously. Provided there is no change in the mass of a gas, the above gas laws can be combined, giving:
For an ideal gas, constant k D mR, where m is the mass of the gas in kg, and R is the characteristic gas constant, i.e.
This is called the characteristic gas equation. In this equation, p D absolute pressure in pascals, V D volume in m3, m D mass in kg, R D characteristic gas constant in J/(kg K), and T D thermodynamic temperature in kelvin.
Some typical values of the characteristic gas constant R include:
air, 287 J/(kg K), hydrogen 4160 J/(kg K), oxygen 260 J/(kg K) and carbon dioxide 184 J/(kg K).
For example, some air at a temperature of 40°C and pressure 4 bar occupies a volume of 0.05 m3. The mass of the air, assuming the characteristic gas constant for air to be 287 J/(kg K), is determined from: pV D mRT, from which, mass of air,
Standard temperature and pressure (i.e. STP) refers to a temperature of 0°C, i.e. 273 K, and normal atmospheric pressure of 101.325 kPa
Kinetic Theory of Gases
The kinetic theory of gases suggests that gases are composed of particles in motion. The continual bombardment of any surface by the gas causes a pressure to be exerted; the greater the density of a gas, the more frequent the number of collisions between molecules and the surface and the greater the pressure exerted. Hence the pressure increases either when the volume of a certain mass of gas is reduced, or when more gas is pumped into a vessel. When the temperature of a gas is increased, the speed of the molecules increases, causing an increase in both the number and the momentum imparted by each collision. This accounts for the increase in pressure of a gas with increase in temperature.
Maxwell (in 1860) explained some of the properties of a gas by assuming that the molecules of a gas make elastic collisions, spend negligible time actually in collision, and themselves occupy a negligible part of the volume of the gas. Also, the attractive forces between molecules are assumed negligible.
It may be shown that for gas occupying a volume V at a pressure p and containing n molecules each of mass m moving at an average velocity of c,
Also, the kinetic energy of the molecules of a gas is proportional to its thermodynamic temperature.
When a liquid evaporates, molecules with sufficient kinetic energy escape from the liquid’s surface. The higher the temperature of the liquid the greater the average kinetic energy of the molecules and the greater the number of molecules that are able to escape. Since it is the molecules with the highest kinetic energy that escape, the average kinetic energy of the remaining molecules decreases and thus the liquid cools.
If a liquid evaporates a vapour is formed. When a vapour exists in the presence of its own liquid a saturated vapour is formed. If all the liquid evaporates an unsaturated vapour is produced. The higher the temperature the greater the number of molecules that escapes to form the vapour. These molecules bombard the walls of the container and thus exert a pressure.
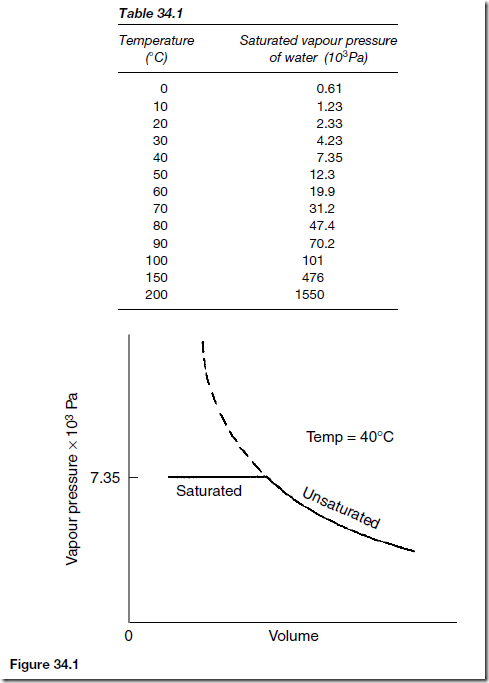
The saturated vapour pressure depends only on the temperature of the vapour. The saturated vapour pressure of water at various temperatures is shown in Table 34.1. A liquid boils at a temperature when its saturated vapour pressure is equal to the atmospheric pressure. Thus water will boil at a temperature greater than 100° C if the atmospheric pressure is increased. This is the principle of the pressure cooker.
A saturated vapour does not obey the gas laws since its pressure depends only on temperature. An unsaturated vapour will obey the gas laws fairly
closely as long as it remains unsaturated. If an unsaturated vapour at a particular temperature is decreased in volume its pressure will rise in accordance with Boyle’s law until it reaches the saturated vapour pressure at that particular temperature (see Figure 34.1). When the vapour pressure at 40° C reaches 7.35 x 103 Pa the vapour becomes saturated as it starts to liquefy.