Magnetic Materials
Magnetic Properties of Materials
The full theory of magnetism is one of the most complex of subjects. How- ever the phenomenon may be satisfactorily explained by the use of a simple model. Bohr and Rutherford, who discovered atomic structure, suggested that electrons move around the nucleus confined to a plane, like planets around the sun. An even better model is to consider each electron as having a surface, which may be spherical or elliptical or something more complicated.
Magnetic effects in materials are due to the electrons contained in them, the electrons giving rise to magnetism in the following two ways:
(i) by revolving around the nucleus
(ii) by their angular momentum about their own axis, called spin.
In each of these cases the charge of the electron can be thought of as moving round in a closed loop and therefore acting as a current loop.
The main measurable quantity of an atomic model is the magnetic moment. When applied to a loop of wire carrying a current,
magnetic moment = current x area of the loop
Electrons associated with atoms possess magnetic moment which gives rise to their magnetic properties.
Diamagnetism is a phenomenon exhibited by materials having a relative permeability less than unity. When electrons move more or less in a spherical orbit around the nucleus, the magnetic moment due to this orbital is zero, all the current due to moving electrons being considered as averaging to zero.
If the net magnetic moment of the electron spins were also zero then there would be no tendency for the electron motion to line up in the presence of a magnetic field. However, as a field is being turned on, the flux through the electron orbitals increases. Thus, considering the orbital as a circuit, there will be, by Faraday’s laws, an e.m.f. induced in it which will change the current in the circuit. The flux change will accelerate the electrons in its orbit, causing an induced magnetic moment. By Lenz’s law the flux due to the induced magnetic moment will be such as to oppose the applied flux. As a result, the net flux through the material becomes less than in a vacuum. Since
materials the relative permeability is less than one.
Paramagnetism is a phenomenon exhibited by materials where the relative permeability is greater than unity. Paramagnetism occurs in substances where atoms have a permanent magnetic moment. This may be caused by the orbitals not being spherical or by the spin of the electrons. Electron spins tend to pair up and cancel each other. However, there are many atoms with odd numbers of electrons, or in which pairing is incomplete. Such atoms have what is called a permanent dipole moment. When a field is applied to them they tend to line up with the field, like compass needles, and so strengthen the flux in that region. (Diamagnetic materials do not tend to line up with the field in this way.) When this effect is stronger than the diamagnetic effect, the overall effect is to make the relative permeability greater than one. Such materials are called paramagnetic.
Ferromagnetic materials
Ferromagnetism is the phenomenon exhibited by materials having a relative permeability which is considerably greater than 1 and which varies with flux density. Iron, cobalt and nickel are the only elements that are ferromagnetic at ordinary working temperatures, but there are several alloys containing one or more of these metals as constituents, with widely varying ferromagnetic properties.
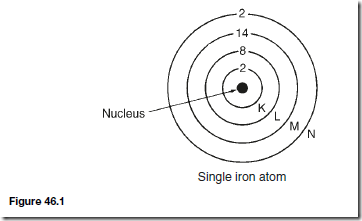
Consider the simple model of a single iron atom represented in Figure 46.1. It consists of a small heavy central nucleus surrounded by a total of 26 electrons. Each electron has an orbital motion about the nucleus in a limited region, or shell, such shells being represented by circles K, L, M and N. The numbers in Figure 46.1 represent the number of electrons in each shell. The outer shell N contains two loosely held electrons, these electrons becoming the carriers of electric current, making iron electrically conductive. There are 14 electrons in the M shell and it is this group that is responsible for magnetism. An electron carries a negative charge and a charge in motion constitutes an electric current with which is associated a magnetic field. Magnetism would therefore result from the orbital motion of each electron in the atom. However, experimental evidence indicates that the resultant magnetic effect due to all the orbital motions in the metal solid is zero; thus the orbital currents may be disregarded.
In addition to the orbital motion, each electron spins on its own axis. A rotating charge is equivalent to a circular current and gives rise to a magnetic field. In any atom, all the axes about which the electrons spin are parallel, but rotation may be in either direction. In the single atom shown in Figure 46.1, in each of the K, L and N shells equal numbers of electrons spin in the clockwise and anticlockwise directions respectively and therefore these shells are magnetically neutral. However, in shell M, nine of the electrons spin in one direction while five spin in the opposite direction. There is therefore a resultant effect due to four electrons.
The atom of cobalt has 15 electrons in the M shell, nine spinning in one direction and six in the other. Thus with cobalt there is a resultant effect due to 3 electrons. A nickel atom has a resultant effect due to 2 electrons. The atoms of the paramagnetic elements, such as manganese, chromium or aluminium, also have a resultant effect for the same reasons as that of iron, cobalt and nickel. However, in the diamagnetic materials there is an exact equality between the clockwise and anticlockwise spins.
The total magnetic field of the resultant effect due to the four electrons in the iron atom is large enough to influence other atoms. Thus the orientation of one atom tends to spread through the material, with atoms acting together in groups instead of behaving independently. These groups of atoms, called domains (which tend to remain permanently magnetised), act as units. Thus, when a field is applied to a piece of iron, these domains as a whole tend to line up and large flux densities can be produced. This means that the relative permeability of such materials is much greater than one. As the applied field is increased, more and more domains align and the induced flux increases.
The overall magnetic properties of iron alloys and materials containing iron, such as ferrite (ferrite is a mixture of iron oxide together with other oxides — lodestone is a ferrite), depend upon the structure and composition of the material. However, the presence of iron ensures marked magnetic properties of some kind in them. Ferromagnetic effects decrease with temperature, as do those due to paramagnetism. The loss of ferromagnetism with temperature is more sudden, however; the temperature at which it has all disappeared is called the Curie temperature. The ferromagnetic properties reappear on cooling, but any magnetism will have disappeared. Thus a permanent mag- net will be demagnetised by heating above the Curie temperature (1040 K for iron) but can be remagnetised after cooling. Above the Curie temperature, ferromagnetics behave as paramagnetics.
Nonpermanent Magnetic Materials
General
Nonpermanent magnetic materials are those in which magnetism may be induced. With the magnetic circuits of electrical machines, transformers and heavy current apparatus a high value of flux density B is desirable so as to limit the cross-sectional area A ( D BA) and therefore the weight and cost
involved. At the same time the magnetic field strength H (=NI/I) should be as small as possible so as to limit the I2R losses in the exciting coils. The relative permeability and the saturation flux density should therefore be high. Also, when flux is continually varying, as in transformers, inductors and armature cores, low hysteresis and eddy current losses are essential.
Silicon-iron alloys
In the earliest electrical machines the magnetic circuit material used was iron with low content of carbon and other impurities. However, it was later
discovered that the deliberate addition of silicon to the iron brought about a great improvement in magnetic properties. The laminations now used in electrical machines and in transformers at supply frequencies are made of silicon-steel in which the silicon in different grades of the material varies in amounts from about 0.5% to 4.5% by weight. The silicon added to iron increases the resistivity. This in turn increases the resistance and thus helps to reduce eddy current loss. The hysteresis loss is also reduced; however, the silicon reduces the saturation flux density.
A limit to the amount of silicon which may be added in practice is set by the mechanical properties of the material, since the addition of silicon causes a material to become brittle. Also the brittleness of a silicon-iron alloy depends on temperature. About 4.5% silicon is found to be the upper practical limit for silicon-iron sheets. Lohys is a typical example of a silicon-iron alloy and is used for the armatures of d.c. machines and for the rotors and stators of a.c. machines. Stalloy, which has a higher proportion of silicon and lower losses, is used for transformer cores.
Silicon steel sheets are often produced by a hot-rolling process. In these finished materials the constituent crystals are not arranged in any particular manner with respect, for example, to the direction of rolling or the plane of the sheet. If silicon steel is reduced in thickness by rolling in the cold state and the material is then annealed it is possible to obtain a finished sheet in which the crystals are nearly all approximately parallel to one another. The material has strongly directional magnetic properties, the rolling direction being the direction of highest permeability. This direction is also the direction of lowest hysteresis loss. This type of material is particularly suitable for use in transformers, since the axis of the core can be made to correspond with the rolling direction of the sheet and thus full use is made of the high permeability, low loss direction of the sheet.
With silicon-iron alloys a maximum magnetic flux density of about 2 T is possible. With cold-rolled silicon steel, used for large machine construction, a maximum flux density of 2.5 T is possible, whereas the maximum obtainable with the hot-rolling process is about 1.8 T. (In fact, with any material, only under the most abnormal of conditions will the value of flux density exceed 3 T).
It should be noted that the term ‘iron-core’ implies that the core is made of iron; it is, in fact, almost certainly made from steel, pure iron being extremely hard to come by. Equally, an iron alloy is generally a steel and so it is preferred to describe a core as being a steel rather than an iron core.
Nickel-iron alloys
Nickel and iron are both ferromagnetic elements and when they are alloyed together in different proportions a series of useful magnetic alloys is obtained. With about 25% – 30% nickel content added to iron, the alloy tends to be very hard and almost nonmagnetic at room temperature. However, when the nickel content is increased to, say, 75% – 80% (together with small amounts of molybdenum and copper), very high values of initial and maximum permeabilities and very low values of hysteresis loss are obtainable if the alloys are given suitable heat treatment. For example, Permalloy, having a content of 78% nickel, 3% molybdenum and the remainder iron, has an initial permeability of 20 000 and a maximum permeability of 100 000 compared with values of 250 and 5000 respectively for iron. The maximum flux density for Permalloy is about 0.8 T. Mumetal (76% nickel, 5% copper and 2% chromium) has similar characteristics. Such materials are used for the cores of current and a.f. trans- formers, for magnetic amplifiers and also for magnetic screening. However, nickel-iron alloys are limited in that they have a low saturation value when compared with iron. Thus, in applications where it is necessary to work at a high flux density, nickel-iron alloys are inferior to both iron and silicon-iron. Also nickel-iron alloys tend to be more expensive than silicon-iron alloys.
Eddy current loss is proportional to the thickness of lamination squared, thus using laminations as thin as possible can reduce such losses. Nickel-iron alloy strip as thin as 0.004 mm, wound in a spiral, may be used.
Dust cores
In many circuits high permeability may be unnecessary or it may be more important to have a very high resistivity. Where this is so, metal powder or dust cores are widely used up to frequencies of 150 MHz. These consist of particles of nickel-iron-molybdenum for lower frequencies and iron for the higher frequencies. The particles, which are individually covered with an insulating film, are mixed with an insulating, resinous binder and pressed into shape.
Ferrites
Magnetite, or ferrous ferrite, is a compound of ferric oxide and ferrous oxide and possesses magnetic properties similar to those of iron. However, being a semiconductor, it has a very high resistivity. Manufactured ferrites are com- pounds of ferric oxide and an oxide of some other metal such as manganese, nickel or zinc. Ferrites are free from eddy current losses at all but the highest frequencies (i.e. >100 MHz) but have a much lower initial permeability com- pared with nickel-iron alloys or silicon-iron alloys. Ferrites have typically a maximum flux density of about 0.4 T. Ferrite cores are used in audio-frequency transformers and inductors.
Permanent Magnetic Materials
A permanent magnet is one in which the material used exhibits magnetism without the need for excitation by a current-carrying coil. The silicon-iron and nickel-iron alloys discussed earlier are ‘soft’ magnetic materials having high permeability and hence low hysteresis loss. The opposite characteristics are required in the ‘hard’ materials used to make permanent magnets. In permanent magnets, high remanent flux density and high coercive force, after magnetisation to saturation, are desirable in order to resist demagnetisation. The hysteresis loop should embrace the maximum possible area. Possibly the best criterion of the merit of a permanent magnet is its maximum energy prod- uct (BH)m , i.e. the maximum value of the product of the flux density B and the magnetic field strength H along the demagnetisation curve (shown as cd in Figure 45.4, page 249). A rough criterion is the product of coercive force and remanent flux density, i.e. (Od)(Oc) in Figure 45.4. The earliest materials used for permanent magnets were tungsten and chromium steel, followed by a series of cobalt steels, to give both a high remanent flux density and a high value of (BH)m Alni was the first of the aluminium-nickel-iron alloys to be discovered, and with the addition of cobalt, titanium and niobium, the Alnico series of magnets was developed, the properties of which vary according to composition. These materials are very hard and brittle. Many alloys with other compositions and trade names are commercially available.
A considerable advance was later made when it was found that directional magnetic properties could be induced in alloys of suitable composition if they were heated in a strong magnetic field. This discovery led to the powerful Alcomex and Hycomex series of magnets. By using special casting techniques to give a grain-oriented structure, even better properties are obtained if the field applied during heat treatment is parallel to the columnar crystals in the magnet. The values of coercivity, the remanent flux density and hence (BH)m are high for these alloys.
The most recent and most powerful permanent magnets discovered are made by powder metallurgy techniques and are based on an intermetallic compound of cobalt and samarium. These are very expensive and are only available in a limited range of small sizes.